
We believe the article would prove to be beneficial in the study of same. So, this is all about the Caesium chemical element to understand the electron configuration and other properties of the element. 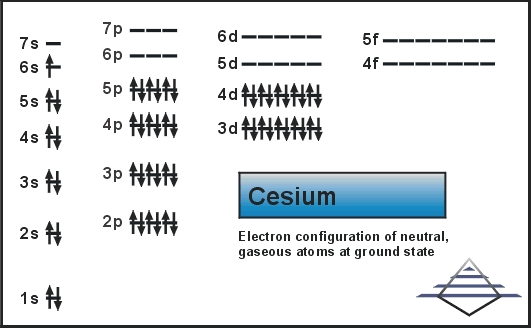
It's outer-most orbital is the 6s1 orbital. It has only one outer s-block electron, however. So, Caesium has a huge role to play in energy production and conversion. Best Answer Copy Cesium has 55 electrons in the neutral atom. The question wants you to recognize the fact that cesium, That is, these elements have one electron located in their outermost shell, i.e their valence shell. This generator does the particular job of converting the heat energy to electricity. Cs is the sixth-period element of group 1 so it has a total of 55 electrons and those electrons can be. So, for the same reason, Caesium remains the highly demanded product in the oil industry.įurthermore, Caesium has other numbers of usages such as in the atomic clocks or in the production of thermionic generators. How to write the Cesium electron configuration. The difference between the mass number of the cesium atom and the number of protons is seventy-eight. The number of neutrons in an atom can be determined by the difference between the atomic mass and the number of protons. It works as the drilling fluid for the same oil extraction purpose. Therefore, a cesium atom has fifty-five protons and fifty-five electrons. The major use of the Caesium goes to the oil industry where the chemical element is used in oil extraction related activities. Ground State Electron Configuration For Cesium. Cesium has eight valence electrons in its outer shell. There is eight valence electron in the outer shell of the cesium. Well, Caesium is a highly practical chemical element that has a number of significant applications. How Many Valence Electrons Does Cesium Have. 
In our case we have cesium (Cs), which is considered as an alkali metal located in the group IA of the periodic table, thus, it has 1 valence electron. For instance, the electron configuration helps in figuring out all the other relevant chemical properties of Caesium or any other element. In the case of groups marked with a letter A next to the Roman number in the periodic table, the Roman numbers determine the number of valence electrons. This configuration of the electron for the chemical element is useful in the further research of the chemical element. This electron distribution equation is what becomes the electron configuration of the element. The electron configuration of Caesium is nothing but the representation of the electrons of the element to its orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
